|
|
| Halite (sodium chloride) mineral.
|
|
|
|
 Fact No. 1 Fact No. 1
Rivers always carry a certain amount of dissolved solids, among them, notably, salt ions.
The amount of total dissolved solids (TDS) varies widely, from less than 100 ppm for some headwater streams
in humid regions, to greater than 1,500 ppm for rivers in arid regions, heavily laden with salts.
For instance, the salinity of the Colorado River at Imperial Dam, California, is about 800 ppm.
|
| Imperial Dam.
| Source: Imperial Irrigation District |
|
 Fact No. 2 Fact No. 2
Dissolved solids in streams and rivers originate in the parent rocks.
| Bioturbation. |
| Source: C. Messina |
|
Weathering and bioturbation are the processes by which the solids are released from the rocks and soils,
leaving the lithosphere and entering
the hydrosphere.
|
 Fact No. 3 Fact No. 3
Several types of dissolved salt cations (positive) and anions (negative) enter the hydrosphere through runoff.
|
The salt cations are four: (1) sodium, (2) calcium, (3) potassium, and (4) magnesium. Typical salt anions are: (1) chloride, (2) sulfate, and (3) carbonate.
| |
| Sodium chloride.
| Source: C. Earley |
|
 Fact No. 4 Fact No. 4
Potassium and magnesium are mostly used by the biosphere, while sodium and calcium are not used by the biosphere
in the amounts in which they are present.
Potassium and magnesium are the good salts, retained by the biosphere.
Sodium and calcium are the bad salts, wasted by the biosphere.
|
 Fact No. 5 Fact No. 5
Evaporation and evapotranspiration leave the waste salts behind, concentrating them in the remaining water bodies.
The amount of total dissolved solids (TDS) in a stream or river increases in the downstream direction.
| |
| Working drain, Imperial valley, California.
|
|
 Fact No. 6 Fact No. 6
Headwater streams have a smaller concentration of dissolved solids than lowland rivers.
Under pristine conditions, the increase in TDS in the downstream direction is a natural characteristic of streams and rivers.
|
| Dissolution of sodium chloride in water.
|
 Fact No. 7 Fact No. 7
Irrigation, a consumptive use of water, produces additional evapotranspiration, increasing the concentration of total dissolved solids in runoff waters.
| Furrow irrigation, Wellton-Mohawk, Arizona.
|
|
Every iota of water used consumptively to grow crops leaves the salts behind, creating a problem of disposal.
|
 Fact No. 8 Fact No. 8
Storage reservoirs produce additional evaporation, increasing the concentration of total dissolved solids in the water body and,
therefore, in the hydrosphere.
Arid regions evaporate water at rates much higher than humid regions; thus, arid regions
concentrate salts faster than humid regions.
|
 Fact No. 9 Fact No. 9
Rivers of developed basins have higher concentrations of TDS than rivers of pristine basins.
| San Joaquin river near Vernalis, California.
| Source: University of California |
|
The additional salts to be mobilized increase the TDS concentration, unless they are routed to and collected in evaporation ponds.
|
 Fact No. 10 Fact No. 10
Evaporation ponds or basins take the extra salts away from the runoff (hydrosphere) and deposit them in the soils (lithosphere).
An evaporation pond eliminates regional surface water
at the cost of increasing local soil/land salinity.
|
| Evaporation pond, Tulare Lake basin, California.
|
|
 Fact No. 11 Fact No. 11
An aquifer lying below an evaporation basin can become contaminated with salts percolating through the vadose zone,
from land surface to groundwater table.
| Vadoze zone. |
Source: U.S. Geological Survey
|
|
A properly designed liner may reduce this effect, but liners have a finite life.
|
 Fact No. 12 Fact No. 12
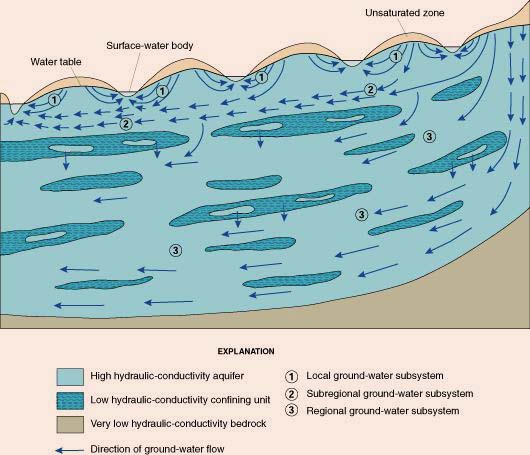
Aquifers flow from zones of higher potential energy
to zones of lower potential energy, in a direction that resembles and/or closely parallels the land surface.
| Direction of groundwater flow.
|
Source: U.S. Geological Survey |
|
The great majority of groundwater eventually makes it to the surface waters through baseflow.
On a global basis, only a very small fraction of groundwater (less than 2%) flows directly into the ocean through deep percolation,
avoiding the surface waters altogether.
|
 Fact No. 13 Fact No. 13
The more intensely a river basin is developed for irrigation, the greater the quantity of
salts that need to be handled or disposed of, either through salt-laden runoff and streamflow (a regional impact),
or by storage in evaporation ponds (a local impact).
|
The choice is either: (1) to pollute the river, or (2) to first pollute the land, and then, eventually, through baseflow, the river. A third choice, to collect the salt and cart it to the ocean,
is usually prohibitively expensive.
| |
| South evaporation pond, Tulare Lake Basin, California.
|
|
|
 Fact No. 1
Fact No. 1
 Fact No. 2
Fact No. 2
 Fact No. 3
Fact No. 3
 Fact No. 4
Fact No. 4
 Fact No. 5
Fact No. 5
 Fact No. 6
Fact No. 6
 Fact No. 7
Fact No. 7
 Fact No. 8
Fact No. 8
 Fact No. 9
Fact No. 9
 Fact No. 10
Fact No. 10
 Fact No. 11
Fact No. 11
 Fact No. 12
Fact No. 12
 Fact No. 13
Fact No. 13