1. A scarce resource Water is a precious and scarce resource. Given the needs of economic development, it is very likely that water will continue to remain a scarce resource in the future. Surface water constitutes only a small fraction of the total amount of fresh water. Therefore, societies are increasingly turning to groundwater to satisfy the demand for fresh water. While the quantity of groundwater pumping is related to the issue of sustainability (Ponce 2007), the quality of groundwater remains a cause for concern. Pumping saline groundwaters has the unintended negative consequence of bringing large quantities of salt to the surface, which require costly and often cumbersome disposal. 2. Geochemistry of natural groundwaters Chebotarev (1955) pioneered the systematic study of the geochemistry of natural groundwaters in the crust of weathering.1 He reckoned that groundwater is never at rest, and that it is always moving along a hydrodynamic gradient from a point of high head to a point of low head (Fig. 1).
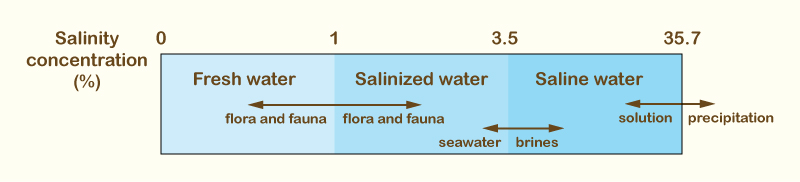
The following ions are present in natural groundwaters in significant quantities: Anions -- Bicarbonate, Sulfate, Chloride; Cations -- Sodium, Potassium, Calcium, and Magnesium. Depending on its salinity concentration, the quality of groundwaters falls into three categories:
The 1% limit separates fresh-water from salt-water flora and fauna; the 3.5% limit is the salinity of seawater; the 35.7% limit is the saturation concentration of sodium chloride in water, at 20oC (Fig. 2).
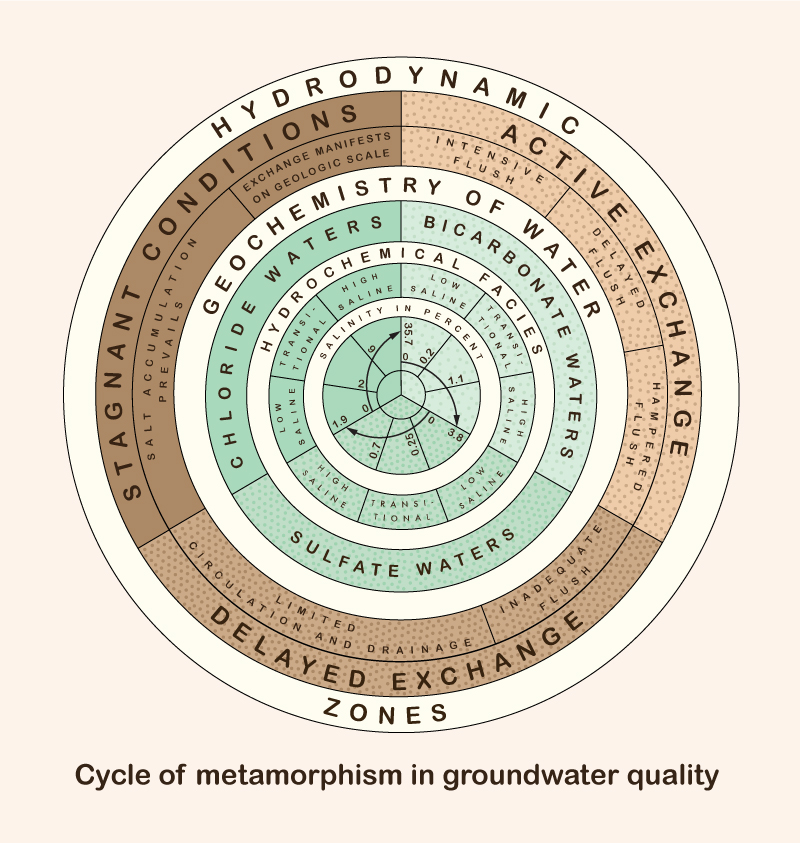
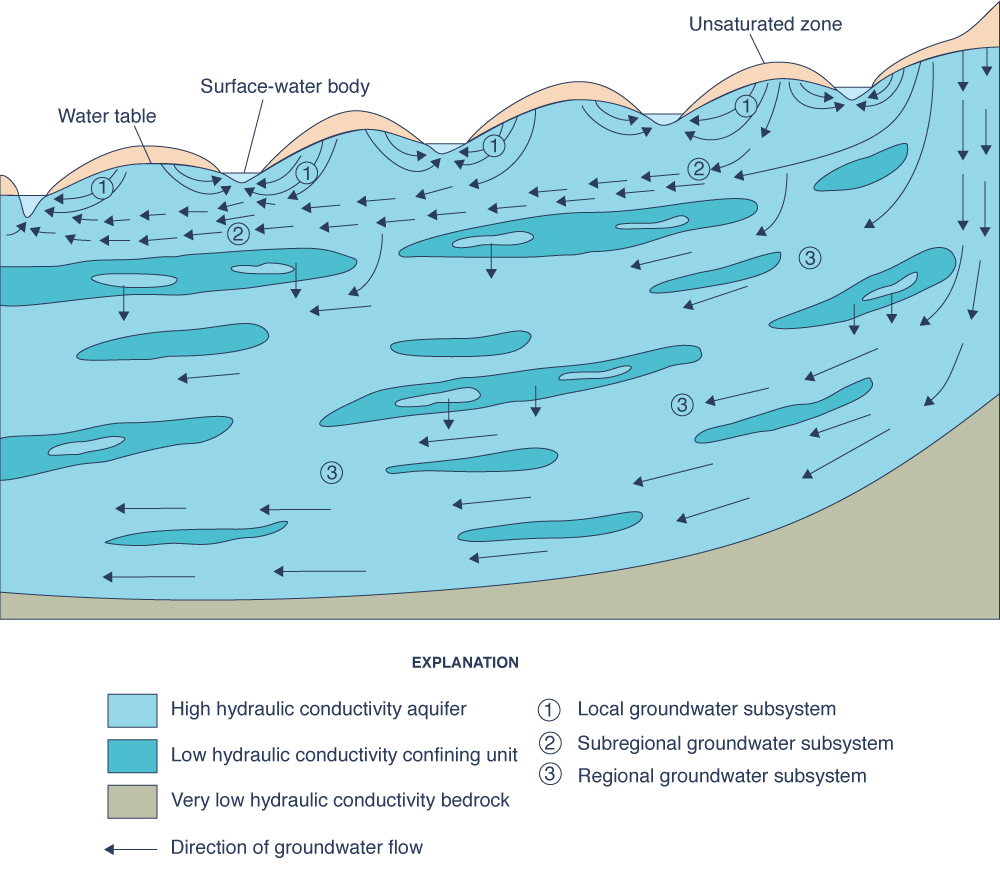
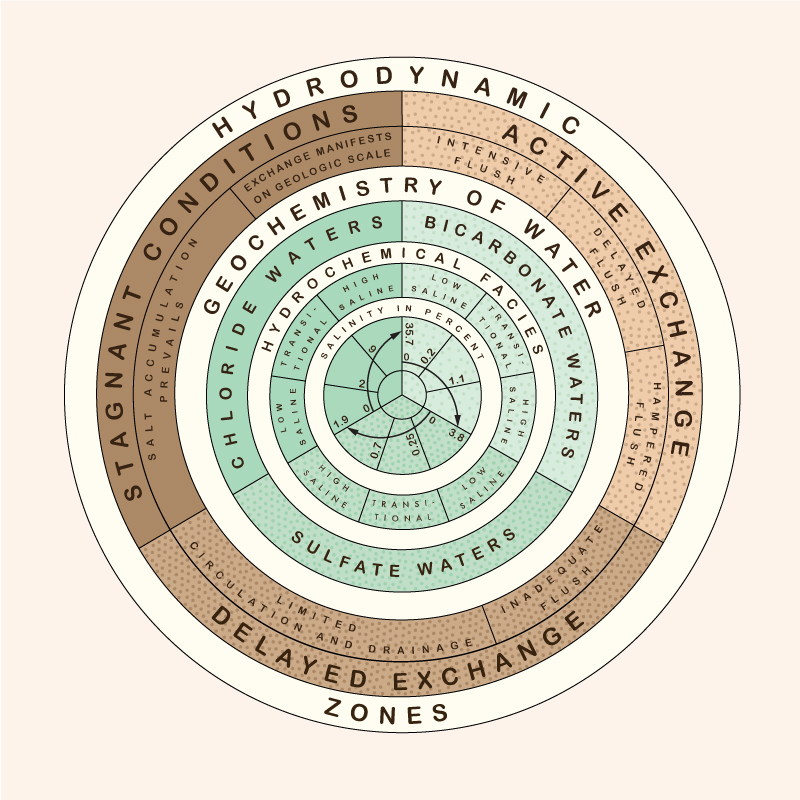
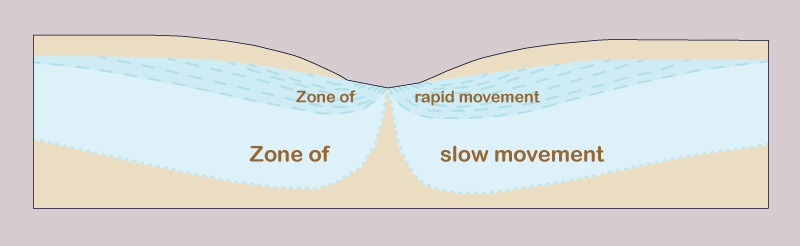
3. Metamorphism of groundwaters Hydrodynamic and hydrochemical gradients affect the distribution and type of groundwaters. There are three hydrodynamic zones in groundwater flow (Fig. 3) (Chebotarev 1955):
The type of exchange, or replenishment rate, is inversely related to the detention time, i.e., locally, the ratio of volume to discharge. Small hydrodynamic gradients lead to low velocities and small discharges and, consequently, to long detention times. The increase in salinity concentration becomes apparent as the exchange rate changes from active to delayed, and eventually to stagnant conditions. Therefore, zones of smaller gradients and slower movement occur in deeper strata (Fig. 4).
The metamorphism of groundwater geochemistry is a function of salinity concentration and depth below the Earth's surface. The following series holds: This is the Chebotarev sequence, expressed in simpler terms as follows: Bicarbonate waters occur near the Earth's surface, while chloride waters occur in the deeper geologic strata. The region of active exchange between these two water types varies widely, sometimes reaching depths of 5,000 to 7,000 ft. The deepest groundwaters belong to the chloride type, at depths over 11,000 ft (2.08 miles), and varying widely. Bicarbonate waters occur at shallower depths, with sulfate waters being of a transitional type. Bicarbonate waters have a salinity between 0 and 3.8%; sulfate waters between 0 and 1.9%; and chloride waters between 0 and 35.7% (Fig. 2). Chemical compounds of higher solubility, sodium, for instance, are found in deeper water in greater relative abundance. Moreover, waters of high salinity have higher specific gravity and, therefore, tend to occupy the lower portions of the water-bearing strata.
4. Salinity gradients in groundwater flow The salinity of groundwaters increases as a function of:
The increase in salinity with distance from the recharge area depends on local conditions. It can be as low as 315 ppm per mile, such as in Minnesota and Wisconsin, or as high as 10,000 ppm per mile, for instance, in the ancient Arbuckle Mountains of Oklahoma (Chebotarev 1955).
The time of travel determines to a large extent the adoption of local geological conditions.
5. Conclusions The salinity of groundwaters is a function of the local water-exchange rate or replenishment rate. As drainage conditions deteriorate with depth, the water-exchange rate is reduced and the salinity concentration increases. The cycle of metamorphism of groundwater types ranges from bicarbonate waters to sulfate waters to chloride waters, as a function of increasing depth, reduced drainage, and longer time of exposure. Pumping groundwater at great depths may have the unintended consequence of bringing large quantities of salt to the surface, where they would have to be disposed of properly, usually at great expense. Therefore, caution is recommended when pumping groundwater at great depths, particularly for consumptive uses such as irrigation. The tradeoffs of using deep groundwater vs bringing excessive amounts of salt to the surface must be evaluated carefully. The overriding objective should be to keep the basin in salt balance by the use of a suitable brine line.2 Examples of brine lines are the Santa Ana River Interceptor (SARI) Line in Orange County, California, and the Calleguas Brine Line, in Ventura County, California. 1 The "crust of weathering" is the upper part of the lithosphere, which consists of the loose products of disintegration of igneous and metamorphic rocks, with a depth of approximately 10 miles (16 km).
2
A brine line is a closed conduit that
conveys a brine or brines from point of generation (inland) to point of disposal, typically an ocean outfall.
|
| 211109 |